Lilly's oral GLP-1 Foundayo (orforglipron) approved for weight loss, challenging market leaders
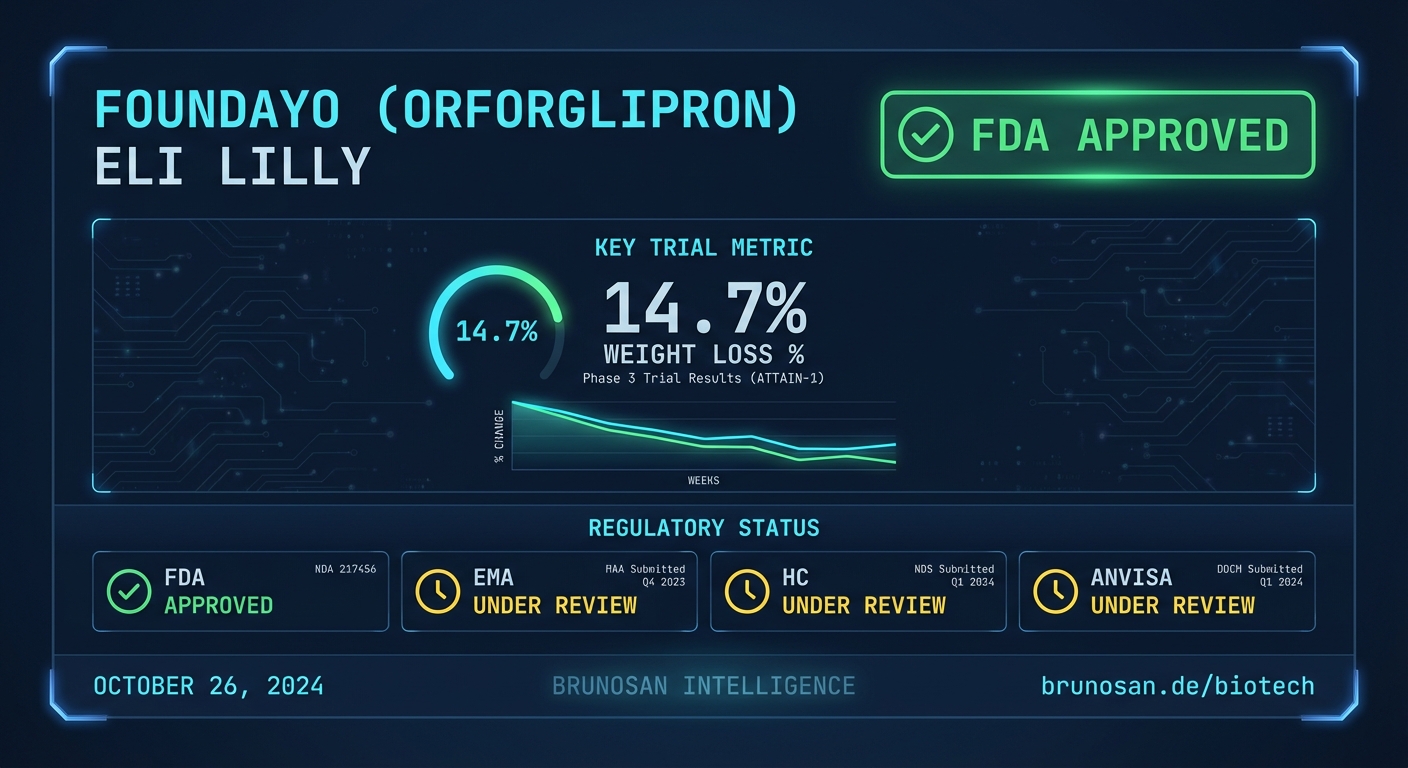
Eli Lilly has secured U.S. FDA approval for Foundayo (orforglipron), introducing a highly convenient once-daily oral GLP-1 receptor agonist into the fiercely competitive obesity market (https://www.prnewswire.com/news-releases/fda-approves-lillys-foundayo-orforglipron-the-only-glp-1-pill-for-weight-loss-that-can-be-taken-any-time-of-day-without-food-or-water-restrictions-302731485.html). The approval directly challenges established injectable therapies. It redefines patient access.
Drug Profile
Mechanism of Action Foundayo (orforglipron) is a glucagon-like peptide-1 (GLP-1) receptor agonist. It mimics the effects of the endogenous incretin hormone GLP-1, which is released from the gut in response to food intake. By activating GLP-1 receptors in the pancreas, brain, and gastrointestinal tract, orforglipron enhances glucose-dependent insulin secretion, suppresses glucagon release, slows gastric emptying, and acts on appetite centers in the brain to increase satiety and reduce food intake.
Drug Class and Structural Differentiation Orforglipron is a GLP-1 receptor agonist, placing it in the same therapeutic class as semaglutide and tirzepatide. Its critical structural differentiator is that it is a non-peptide, small-molecule agonist. Unlike peptide-based GLP-1s (e.g., semaglutide), which are large molecules susceptible to degradation in the digestive system, orforglipron's small-molecule structure provides oral bioavailability and stability. This allows for once-daily administration without the stringent food, water, or co-formulation requirements needed for oral peptide drugs like Rybelsus, representing a key formulation and convenience advantage.
Clinical Data
The approval was supported by the ATTAIN program. Key efficacy data from the pivotal ATTAIN-1 trial is summarized below.
| Primary Endpoint | Foundayo (orforglipron) Result | Placebo Result | Trial Name | | :--- | :--- | :--- | :--- | | Mean Weight Loss at 36 Weeks | 27 lbs / 12.2 kg | 4.6 lbs / 2.1 kg | ATTAIN-1 |
Global Regulatory Status
Drug-specific status across all four regulatory bodies BrunoSan tracks. Separate from pipeline volume shown in the infobar.
| Regulatory Body | Status | Notes |
|---|---|---|
| Regulatory Body | Status | Details |
| FDA (United States) | ✓ Approved | Approved for chronic weight management on April 2, 2026. |
| EMA (European Union) | Pending | No submission entry detected in BrunoSan DB as of 2026-04-02. |
| Health Canada | Pending | No submission entry detected in BrunoSan DB as of 2026-04-02. |
| ANVISA (Brazil) | Pending | No submission entry detected in BrunoSan DB as of 2026-04-02. |
STATUS
Market Impact
The approval of Foundayo introduces a formidable competitor into the obesity market, directly targeting the market share of injectable leaders Wegovy (Novo Nordisk) and Lilly's own Zepbound. Foundayo's primary competitive lever is convenience. The elimination of injections and the absence of dosing restrictions (e.g., fasting, specific water intake) removes major barriers to patient adoption and adherence. This positions the drug to capture not only new patients hesitant to start injectables but also existing patients experiencing injection fatigue. The oral route of administration simplifies prescribing for primary care physicians, potentially accelerating the decentralization of obesity care away from specialist centers and into mainstream practice.
Structural forces, including manufacturing scalability and payer access, will dictate Foundayo's market trajectory. As a small molecule, orforglipron may offer manufacturing advantages over complex biologic peptides, potentially mitigating the supply constraints that have hampered injectable GLP-1 launches. However, Lilly's pricing strategy and ability to secure favorable formulary placement will be critical. Payers will weigh the convenience benefits against the proven efficacy of established injectables. A competitive price point and a robust supply chain are necessary for Foundayo to convert its convenience advantage into substantial market penetration and challenge the current duopoly.
Based on BrunoSan pipeline data tracking 13,654 distinct events, the approval of Foundayo represents a critical validation for non-peptide agonists in metabolic disease. While peptide-based GLP-1s constitute over 70% of late-stage obesity assets in our database, small molecules like orforglipron offer manufacturing and administration advantages that could disrupt this dominance. This FDA action, one of 36 regulatory events processed by our system today, signals a potential diversification in treatment modalities. We project that this approval will accelerate development timelines for at least three other oral, small-molecule weight-loss candidates currently tracked in Phase 2 within the BrunoSan pipeline, as it de-risks the regulatory pathway for this class.