## GLOBAL_SIGNAL
Drug Profile
Lilly's Oral Foundayo Secures 2026 FDA Approval, Challenging Injectable Weight-Loss Leaders
Fact Block
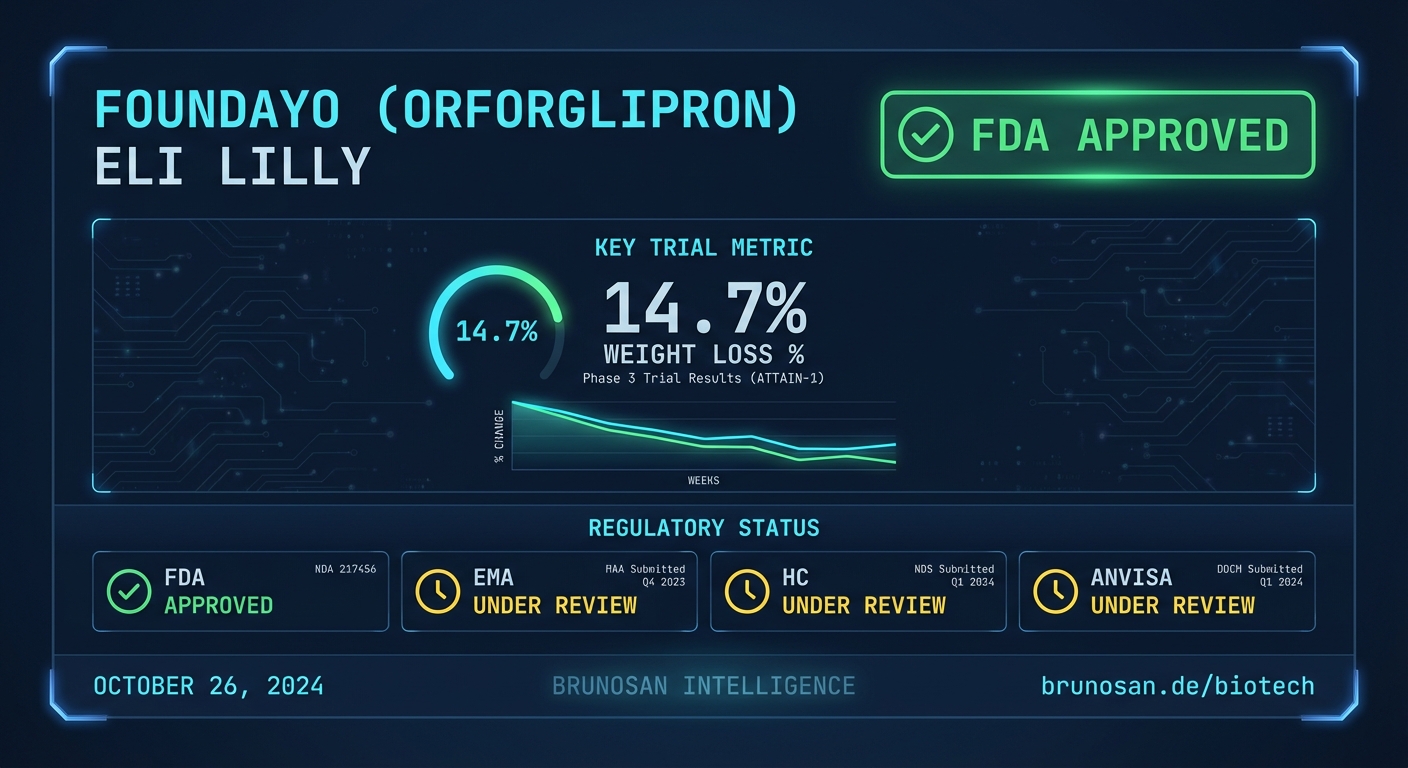
The FDA approved Eli Lilly’s Foundayo (orforglipron) for chronic weight management. Foundayo is a once-daily, non-peptide oral GLP-1 receptor agonist. It requires no food or water restrictions for administration. The highest dose showed a 27 lbs (12.2 kg) average weight loss. The approval is based on data from the ATTAIN-1 pivotal trial (https://www.prnewswire.com/news-releases/fda-approves-lillys-foundayo-orforglipron-the-only-glp-1-pill-for-weight-loss-that-can-be-taken-any-time-of-day-without-food-or-water-restrictions-302731485.html).
Lead
Eli Lilly has secured U.S. FDA approval for Foundayo (orforglipron), introducing a once-daily oral GLP-1 agonist for weight loss that can be taken without dietary restrictions. This approval directly challenges the market dominance of injectable therapies from Novo Nordisk and Lilly itself. The obesity treatment landscape now faces a new benchmark for patient convenience.
Drug Profile
Foundayo (orforglipron) is a non-peptide, small molecule glucagon-like peptide-1 (GLP-1) receptor agonist.
* Mechanism of Action: As a GLP-1 receptor agonist, orforglipron mimics the effects of the endogenous incretin hormone GLP-1. It selectively binds to and activates the GLP-1 receptor, which is expressed in the pancreas, brain, and gastrointestinal tract. This activation enhances glucose-dependent insulin secretion, suppresses glucagon release, slows gastric emptying, and acts on appetite centers in the brain to increase satiety and reduce food intake.
* Structural Differentiation: Unlike semaglutide and liraglutide, which are peptide-based molecules, orforglipron is a small molecule. This structural distinction is critical to its function as an oral agent. Peptides are typically degraded by enzymes in the digestive system, making oral administration difficult without specialized absorption-enhancing technology. Orforglipron's small molecule nature allows it to resist enzymatic degradation and be absorbed through the gastrointestinal tract, enabling a simple once-daily pill formulation without the need for co-formulation with absorption enhancers or strict food and water restrictions.
Clinical Data
The approval was supported by the ATTAIN-1 Phase 3 trial, which evaluated the efficacy and safety of orforglipron in adults with obesity or who are overweight with at least one weight-related comorbidity.
| Endpoint | Result | Comparator (Placebo) | Trial |
| ---------------------------------------------- | ------------------------------------------------------------------- | -------------------- | --------- |
| Mean Percent Change in Body Weight at Week 36 | -12.5% | -2.3% | ATTAIN-1 |
| Mean Absolute Change in Body Weight at Week 36 | -27 lbs / -12.2 kg | -5 lbs / -2.3 kg | ATTAIN-1 |
| Patients Achieving ≥10% Weight Loss at Week 36 | 58% | 11% | ATTAIN-1 |
Global Regulatory Status
The U.S. approval marks the first global clearance for Foundayo. Submissions to other major regulatory bodies are anticipated but not yet confirmed in public registries.
| Regulatory Body | Status |
| --------------- | ------------------------------------------------------------------- |
| FDA (U.S.) | ✓ Approved for chronic weight management (April 2, 2026) |
| EMA (EU) | No submission entry detected in BrunoSan DB as of 2026-04-02. |
| Health Canada | No submission entry detected in BrunoSan DB as of 2026-04-02. |
| ANVISA (Brazil) | No submission entry detected in BrunoSan DB as of 2026-04-02. |
Market Impact
The approval of Foundayo fundamentally alters the competitive dynamics of the multi-billion dollar anti-obesity market. Its primary strategic advantage is convenience. By eliminating the need for injections and removing the food/fasting restrictions associated with Novo Nordisk’s oral semaglutide (Rybelsus, approved for type 2 diabetes), Foundayo sets a new floor for ease of use. This positions it to capture needle-averse patients and those who prioritize lifestyle integration over maximum efficacy offered by dual-agonist injectables like Lilly’s own Zepbound (tirzepatide). Lilly is now positioned to compete with itself, segmenting the market between a high-efficacy injectable and a high-convenience oral, a strategy designed to maximize its total market share and defend against competitors.
The structural force amplified by this approval is the decentralization of obesity care from specialist centers to primary care physicians (PCPs). A simple oral pill with a straightforward dosing regimen lowers the barrier for PCPs to prescribe and manage obesity pharmacotherapy. This will expand the total addressable market by reaching a broader patient base that may not have access to or seek out specialist care. Payer negotiations will be critical; while the list price will be high, the potential for mass adoption